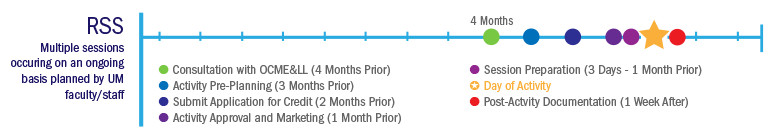
Regularly Scheduled Series (RSS) Planning Timeline
(e.g., Grand Rounds, Tumor Boards, M&M Conferences, Quarterly Meetings)
What is an RSS?
An educational activity with multiple sessions occurring on an ongoing basis (i.e., weekly, monthly, quarterly).
Note: This page can be printed and used as a checklist throughout the planning process
◉ Consult with Office of CME & Lifelong Learning: 4 Months Prior
Initial consultation and assessment to determine if the series aligns with departmental and institutional priorities, and that sufficient time is available for planning, approval, and promotion.
NOTE: The application requires information from three (3) previously held sessions to illustrate the educational planning across the series as a whole. The application cannot be submitted without this information.
Key tasks include:
| Educational Planner | OCME&LL |
| ▢Identify Educational Planner | ▢Participate in consultation with Educational Planner |
| ▢Review activity timeline | |
| ▢Review Planning Guide | |
| ▢Read Designing an RSS that Effectively Translates New Knowledge into Practice | |
| ▢Determine if other credits will be offered | |
| ▢Consult with Department Chair for pre-approval and financial resources |
◉ Activity Pre-Planning: 3 Months Prior
Allocate resources to the activity.
Key tasks include:
| Educational Planner | OCME&LL |
| ▢Identify a Co-planner and/or Planning Committee | |
| ▢Designate an Activity Coordinator | |
| ▢Review the Mitigating Commercial Bias process | |
| ▢If the Educational Planner has any financial relationships, the OCME-LL Financial Relationship Mitigator will be notified | |
| ▢Establish frequency, day, time, and location |
◉ Submit Application for Credit: 2 Months Prior
The application facilitates and documents the planning process.
Key tasks include:
| Educational Planner | OCME&LL |
| ▢Identify current problems in practice, underlying educational needs, and learning objectives | ▢Confirm receipt of application |
| ▢Identify potential presenters and topics | |
| ▢If jointly provided with another organization, develop budget | |
| ▢Collect information on financial relationships | |
| ▢Develop a method for evaluating the activity | |
| ▢Create promotional materials | |
| ▢Submit CME application |
◉ Activity Approval and Marketing: 1 Month Prior
OCME&LL staff reviews and approves the CME application.
Key tasks include:
| Educational Planner / Activity Coordinator | OCME&LL |
| ▢Create activity web page | ▢Begin review of application within 2 business days of submission |
| ▢Request session materials from faculty | ▢Train Activity Coordinator on documentation requirements and how to register sessions in MiCME™ |
| ▢Distribute promotional materials to target audience | ▢If CME fees apply, invoice department |
◉ Session Preparation and Delivery: 3 Days to 1 Month Prior
Final logistical arrangements are made.
Key tasks include:
| Faculty Planner / Activity Coordinator | OCME&LL |
| ▢Collect final faculty presentations |
▢Ensure all relevant financial relationships have been successfully managed |
| ▢Register session for credit in MiCME™ | |
| ▢Assemble participant materials/handouts | |
| ▢Secure on-site staff support as needed |
★ Session Delivery: Day of Activity
CME announcements are made.
Key tasks include:
| Educational Planner | OCME&LL |
| If done for each session rather than annually: | |
| ▢Share information with participants regarding any financial relationships (including if none reported) before each session | |
| ▢Inform participants how to evaluate the activity and how to claim credit | |
| ▢If commercial support is received, share information regarding all support with participants before each session begins (Activity Information Sheet) |
◉ Post Session: 1 Week After
Post-session documentation is submitted to OCME&LL within 7 days of each session of the series.
NOTE: If satisfactory documentation is not received by OCME&LL, future CME credit will not be awarded.
Key tasks include:
| Educational Planner / Activity Coordinator | OCME&LL |
| ▢If done for each session rather than annually: Submit documentation that financial relationship information was shared with participants (including "This speaker has no relevant financial relationships") | ▢Review COI documentation shared with participants for compliance |
| ▢Advise Educational Planner and Activity Coordinator as needed |


